Molarity, Molality, Volume & Mass Percent, Mole Fraction & Density - Solution Concentration Problems - YouTube

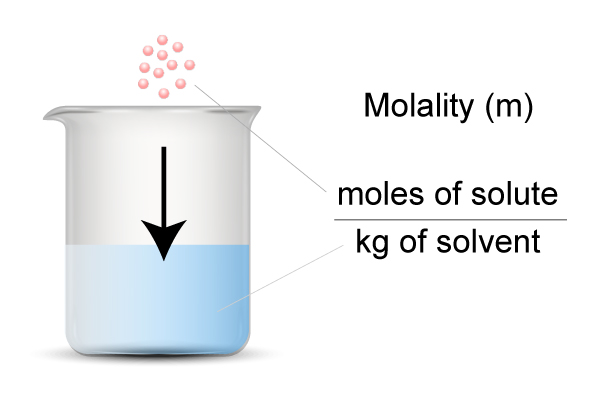
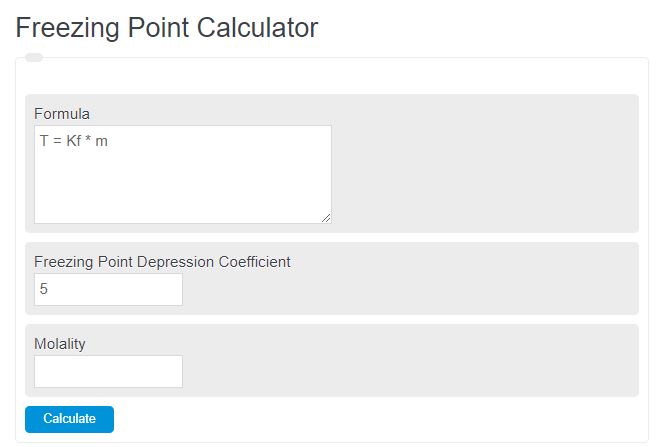
Welcome to Chem Zipper.com......: Calculate the molality and Molarity of a solution of H2SO4 that is 49 % (w/W) and has a density of 1.1g/cc